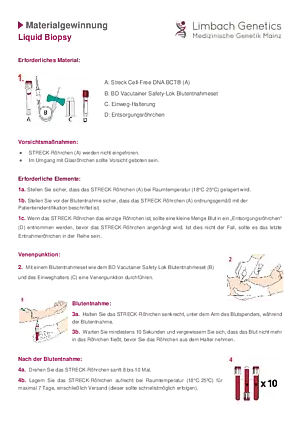
A liquid biopsy is used to analyze circulating tumor markers that can be found in body fluids. These markers include circulating tumor DNA (ctDNA). During cell death and other physiological processes, cells release DNA fragments (cfDNA, cell-free DNA), which can be detected in plasma, among other things. If a tumor disease is present, ctDNA can be detected as a fraction of cell-free DNA. It can be examined using molecular genetics and differentiated from the cell-free DNA of healthy cells using tumor-specific characteristics.
Great potential for the diagnosis of solid tumors
For the diagnosis of solid tumors, ctDNA analysis with liquid biopsy offers wide-ranging potential in various areas of application that are currently being further evaluated in research. If the tumour is molecularly characterized, therapies can be selected in a more targeted manner. Monitoring during therapy makes it possible to assess the response to treatment and identify possible resistance. The procedure also supports the early detection of recurrence and the determination of minimal residual disease (MRD). This also applies to breast cancer, as shown by promising study results (1) for various of the fields of application mentioned.
Minimally invasive and with high information content
- good possibility for serial sample collection thanks to minimally invasive sampling
- No limitation due to availability of biopsy material, as body fluids are analyzed
- Tumor is imaged in all its heterogeneity
- possibility of differentiating between driver and passenger mutations
- Monitoring of clonal development is possible with repeated sampling
- High correlation between ctDNA level and tumor burden
Already part of routine care today
With the EMA (European Medicines Agency) approval of the targeted therapeutics alpelisib (PIK3CA inhibitor) and elacestrant (selective estrogen receptor degrader), liquid biopsy has found its way into companion diagnostics in the care of patients with breast cancer. Both agents are indicated in post-menopausal women and in men with locally advanced or metastatic estrogen receptor (ER)-positive, HER2-negative breast cancer.
Patients with progression during or after anti-hormone therapy and PIK3CA mutation show improved progression-free survival with combination therapy of alpelisib with the anti-oestrogen fulvestrant compared to placebo + fulvestrant(2) According to the recommendation of the European Society of Medical Oncology (ESMO), the PIK3CA mutation can be detected by liquid biopsy or in biopsies(3).
An activating mutation in the estrogen receptor 1 gene ESR1 can be acquired during treatment with aromatase inhibitors. It causes constitutive activation of the oestrogen receptor and thus endocrine resistance. In the phase III EMERALD trial, treatment with elacestrant, which induces the degradation of the oestrogen receptor, improved progression-free survival with ESR1 mutation compared to a standard-of-care approach(4). Elacestrant is indicated in cases of non-response or progression after at least one endocrine therapy, including a CDK4/6 inhibitor, and detection of an activating mutation in ESR1 (preferably by liquid biopsy)(3).
Significant added value for patient care
We use a highly sensitive next-generation sequencing (NGS) method for liquid biopsy, which is even CE-certified as an in-vitro diagnostic tool. For the PIK3CA and ESR1 genes, we are not limited to hotspot mutations, but can also detect other therapy-relevant mutations. By using error correction methods, a very high sensitivity with a detectability of up to 0.06 % VAF (variant allele frequency) can be guaranteed.
PCR-based methods have significant limitations, as they can only be used to specifically analyze well-defined, known variants. For mutation detection, we therefore only test for the most common PIK3CA and ESR1 mutations. We therefore rely on NGS sequencing.
Versatile and future-proof
In addition to target regions in PIK3CA and ESR1, our liquid biopsy approach covers other genes relevant to breast cancer: AKT1, ERBB2, KRAS and TP53. Internal calibration also makes it possible to quantify the number of molecules. In this way, we achieve versatility with our approach, thanks to which we can offer sensitive companion diagnostics for breast cancer particularly quickly in the future expansion of liquid biopsy applications in clinical routine.
Sources
(1) Dickinson et al. JAMA Netw Open 7 (9), e2431722 (2024), Guo et al. J Natl Cancer Cent 4 (1), 63-73 (2024), Guo et al. J Natl Cancer Cent 4 (2), 153-161 (2024), Nader-Marta et al. ESMO Open 9 (3), 102390 (2024), Panet et al. NPJ Breast Cancer 10 (1), 50 (2024)
(2) André et al. N Engl J Med 380 (20),1929-1940 (2019), André et al. Ann Oncol 32 (2), 208-217 (2021)
(3) Pascual et al. Ann Oncol 33 (8), 750-768 (2022)
(4) Bidard et al. J Clin Oncol 40, 3246-3256 (2022), Burstein et al. J Clin Oncol 41 (18), 3423-3425 (2023)